This website uses cookies to improve your experience. We'll assume you're ok with this, but you can opt-out if you wish. Mehr erfahren
Category: Forschung

The ALS app at the international ALS symposium in San Diego
On December 4, 2025, the ALS app will be presented at the“ALS App Europe Initiative Meeting” at the International Symposium on ALS/MND in San Diego (USA). The meeting brings together European and American ALS centers, researchers and patient initiatives. You can find out more about the program here: https://symposium.mndassociation.org/programme/satellite-meetings/ The “ALS App Europe Initiative” meeting…

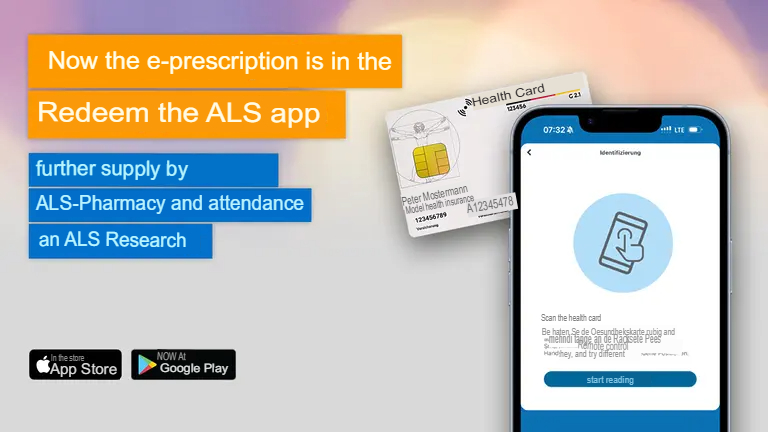
The new ALS-OPM classification is now in the ALS app.
We are pleased to introduce an important new feature in the ALS app today: The new OPM classification is now live! The OPM classification has been available in the current app version 5.10.4 for iOS (Apple) and Android (Google) since yesterday. What does the OPM classification mean? The phenotype of ALS – i.e. the individual…

Patient survey on eye control glasses for people with ALS
The patient survey on eye control glasses is a nationwide, systematic survey of patients with amyotrophic lateral sclerosis (ALS). The aim of the survey is to gain important insights into expectations of the eye-control glasses as well as experiences and satisfaction following treatment with the innovative aid. Drive the wheelchair safely with eye control glasses…

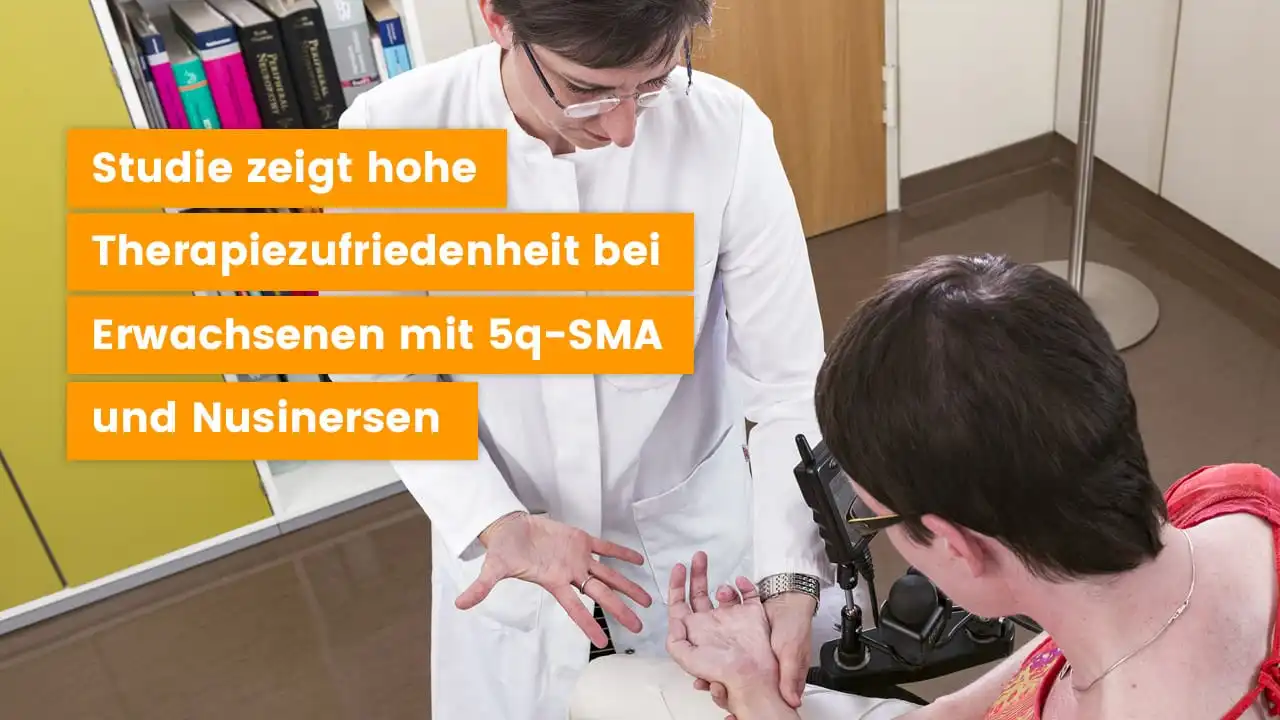
Study on the treatment of people with 5q-SMA with nusinersen in adulthood
Treatment with the drug nusinersen is available for people with 5q-associated spinal muscular atrophy (5q-SMA). In our application study with people with 5q-SMA who received nusinersen therapy in adulthood, the treatment goals, symptom progression and treatment satisfaction with the drug were systematically investigated. In the period from July 2019 to September 2020, Ambulanzpartner conducted an…

The SMA app during the coronavirus pandemic: support for adults with SMA and doctors
The corona pandemic necessitates infection control measures that are associated with a significant change in the care and treatment of people with spinal muscular atrophy (SMA). In this situation, telemedical visits between doctors and patients take on a previously unknown significance. Digital recording of SMA symptoms During a telemedical visit, the systematic communication of symptoms…
Study to record treatment with nusinersen in adults
Nusinersen (Spinraza®) is approved for the treatment of 5q-associated spinal muscular atrophy (SMA). For infants and children, nusinersen can halt the progression of the disease and improve muscle function. For adults, no systematic data on treatment with nusinersen are available to date. Scientific treatment documentation is therefore necessary to improve the data situation for adults…