The nusinersen application study is a nationwide, systematic patient survey of 100 patients with spinal muscular atrophy (SMA), in which important findings on the efficacy of nusinersen in adulthood are gained.
The drug Nusinersen
The drug nusinersen (trade name Spinraza) was approved for the treatment of SMA in Europe in 2017 on the basis of two clinical trials in children (CHERISH and ENDEAR trials). The genetic form of SMA is caused by a mutation in the SMN1 gene, which plays an important role in the development and maintenance of motor function. Adults with SMA could not be included in the approval studies, which is why only very limited study data on the efficacy of nusinersen is available. The approval and reimbursement of nusinersen therapy in adults is based on the assumption that the disease process is comparable in children and adults.
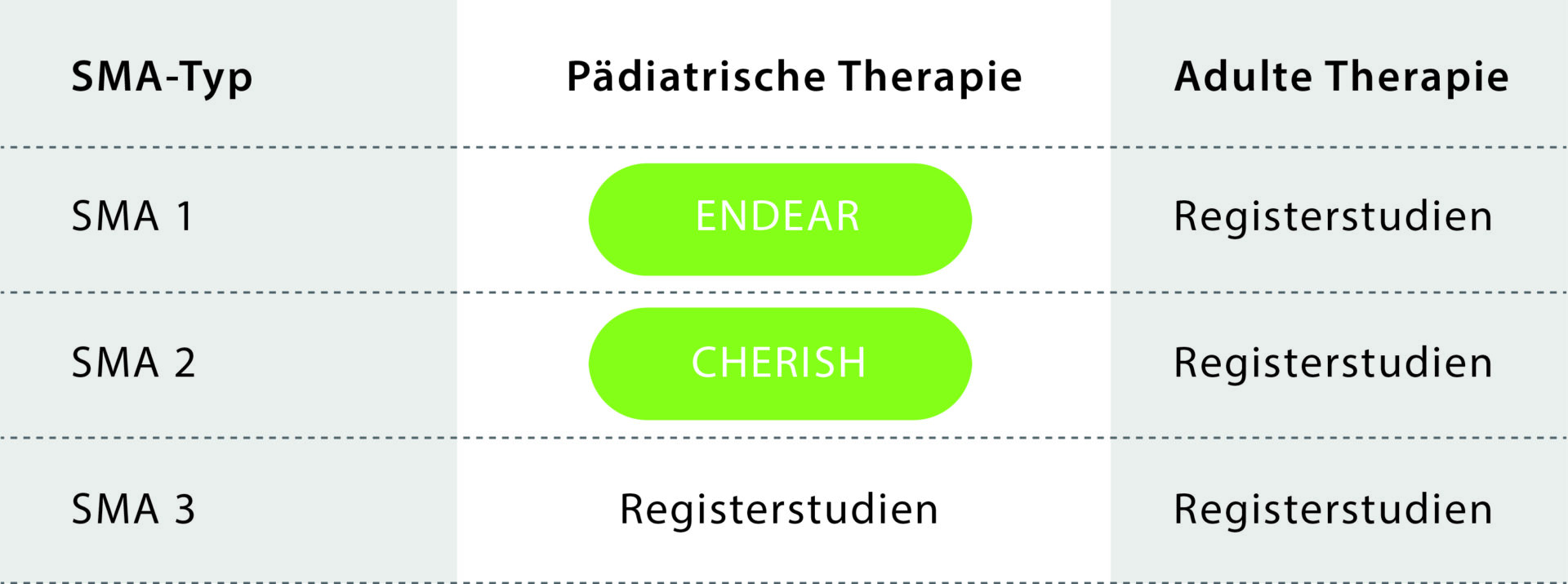
Figure – Study situation for nusinersen in SMA: Studies on the efficacy of nusinersen were conducted in children (“pediatric therapy”) with SMA types 1 and 2 (disease in the first 18 months of life). No clinical studies were conducted for SMA type 3 (disease > 18 months of life) and for patients in adulthood (all SMA types). Registry studies are therefore being conducted to evaluate the benefits of nusinersen for adult SMA patients (“adult therapy”).
The Nusinersen application study
In Germany, the Federal Joint Committee (G-BA) is responsible for the evaluation (and recommendation of health insurance reimbursement) of drugs. The G-BA has determined a benefit of nusinersen for SMA and refers to the approval studies with children. For the benefit assessment in adults with SMA, there is a “knowledge gap” that is to be closed by various registry studies (such as the nusinersen application study).
In connection with the benefit assessment of nusinersen, the following research questions are to be clarified within the framework of the application study:
– What is the age spectrum and distribution of SMA progression forms (SMA type 1-3) with Nusinersen therapy?
– What motor deficits exist at the start of Nusinersen therapy?
– What expectations do people with SMA have of Nusinersen treatment and what influence do age, gender or SMA type have?
– Is there a change in SMA symptoms (improvement, cessation, worsening) as a result of Nusinersen therapy?
– Is there a correlation between patient age, duration of illness and efficacy?
– What is the recommendation rate and patient satisfaction with the drug and the treatment circumstances?
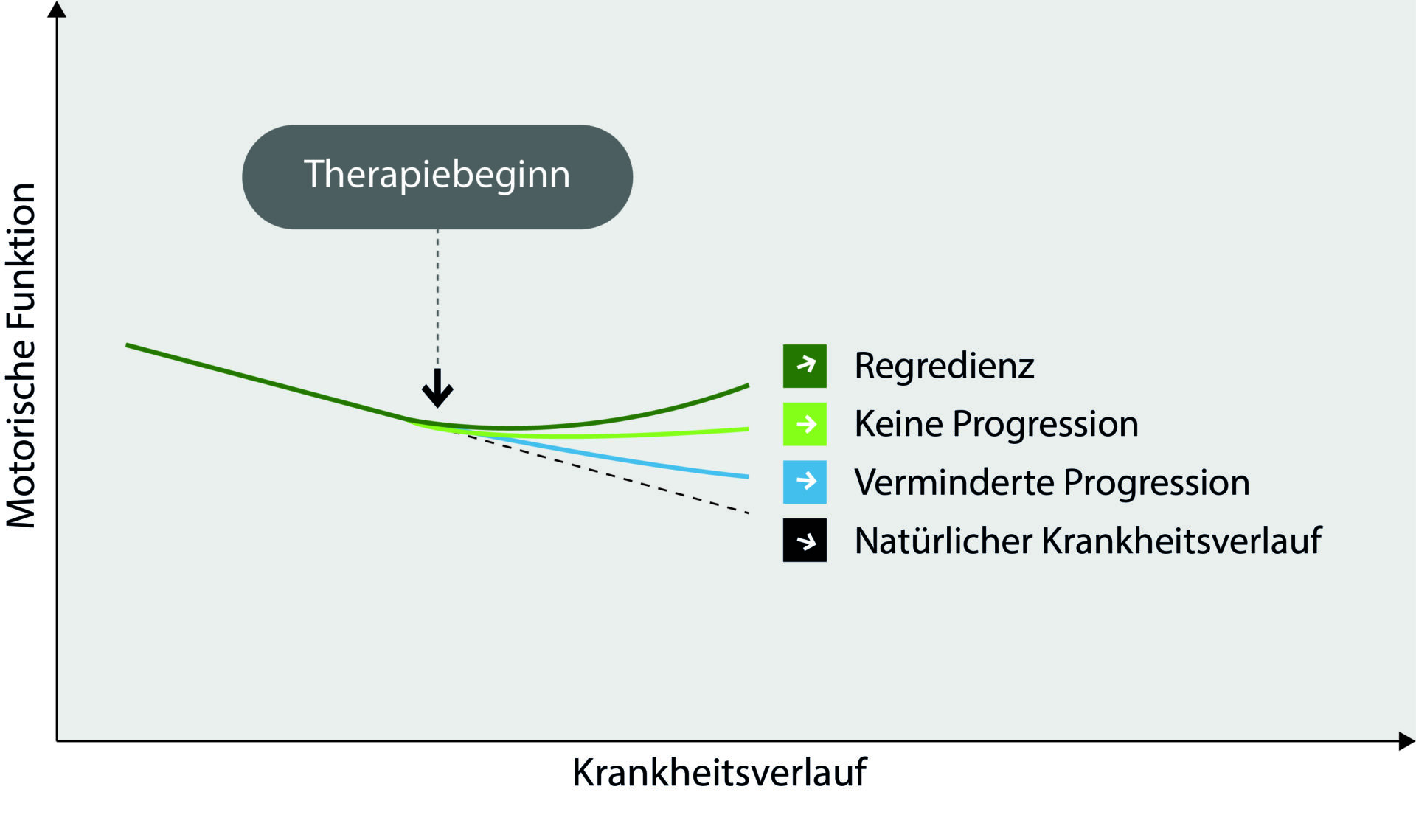
Figure – Possibilities of nusinersen efficacy: In the therapeutic effect of nusinersen, there are basically 4 possibilities in the course of the disease
: 1) no effect (“natural course of the disease”); 2) slowing (“reduced progression”); 3) standstill (“no progression”);
4) improvement (“regression”).
Participation in the Nusinersen application study
The following criteria must be met in order to participate in the study:
– Minimum age: 18 years
– Diagnosis of SMA with mutation in the SMN1 gene (5q-SMA)
– Ongoing or planned therapy with nusinersen (start of treatment in the next 3 months)
– Consent to the registry study and to data collection on the outpatient partner research platform
Procedure of the Nusinersen application study
The application study is carried out in the form of a comprehensive survey. The questionnaire can be completed in different ways:
– Sending by post, independent completion of the questionnaire and return with stamped envelope
– Survey during the Nusinersen clinic stay
– Visit at home with personal survey
– Telephone appointment and telephone survey
– Online survey via the outpatient partner research platform
Duration and scope of the survey
Answering all questions takes around 90 to 120 minutes and will take place every 4 months until June 2021.
Funding of the Nusinersen application study
The study is supported by the research-based pharmaceutical company Biogen GmbH. Biogen is responsible for the development and production of the SMA drug.
Organization and implementation of the Nusinersen application study
The study is being carried out by ambulance partner Soziotechnologie APST GmbH (APST) in cooperation with nine SMA centers in Germany. APST is responsible for organizing the study and providing a digital research platform. The study has been approved by the ethics committee of Charité – Universitätsmedizin Berlin.
Study centers of the Nusinersen application study
The following nine SMA centers are participating in the study:
– Charité – Universitätsmedizin Berlin – Outpatient Clinic for ALS
Augustenburger Platz 1, 13353 Berlin
– University Hospital Bergmannsheil – Outpatient Clinic for Motor Neuron Diseases
Bürkle-de-la-Camp-Platz 1, 44789 Bochum
– University Hospital Bonn – Clinic for Neurodegenerative Diseases
Sigmund-Freud-Straße 25, 53127 Bonn
– University Hospital Carl Gustav Carus Dresden – Outpatient Clinic for Motor Neuron Diseases
Fetscherstrasse 74, 01307 Dresden
– University Hospital Essen – Outpatient clinic for ALS
Hufelandstraße 55, 45147 Essen
– Hannover Medical School – Outpatient Clinic for ALS
Carl-Neuberg-Straße, 130625 Hannover
– Heidelberg University Hospital – Neuromuscular Outpatient Clinic
Im Neuenheimer Feld 400, 69120 Heidelberg
– University Hospital Jena – Outpatient clinic for ALS
Erlanger Allee 101, 07747 Jena
– University Medicine Rostock, ALS Outpatient Clinic
Gehlsheimer Straße 20, 18147 Rostock
Use of the results of the Nusinersen application study
The results are made available to Biogen in anonymized form and without identifying individuals. This data is of great importance for the establishment and further development of nusinersen therapy. It is planned to include the study results in the benefit assessment by the G-BA. In addition, the results will be published at scientific congresses and in international journals. Summaries of interim or final analyses will be sent in the form of a newsletter to SMA patients who are registered on the outpatient partner platform and have agreed to receive newsletters.
Contact us
Study telephone: 0151 – 648 406 30
Head office: 030 – 810 314 10
Mail: nusinersen@ambulanzpartner.de
This website uses cookies to improve your experience. We'll assume you're ok with this, but you can opt-out if you wish. Read More

