Treatment with the drug nusinersen is available for people with 5q-associated spinal muscular atrophy (5q-SMA). In our application study with people with 5q-SMA who received nusinersen therapy in adulthood, the treatment goals, symptom progression and treatment satisfaction with the drug were systematically investigated.
In the period from July 2019 to September 2020, Ambulanzpartner conducted an application study at 9 SMA treatment centers in Germany. The data of 151 people with the following inclusion criteria were recorded and analyzed:
1) Diagnosis of a 5q-SMA
2) Treatment with Nusinersen
3) Registration on the ambulance partner research platform

Figure: SMA treatment centers participating in the Nusinersen application study.
The results of this scientific data analysis were published in May in the renowned neurological journal European Journal of Neurology. To the article: https://onlinelibrary.wiley.com/doi/10.1111/ene.14902
Wide range of ages, duration of illness and motor-functional deficits
There were slightly more men (56%, n = 84) than women (44%, n = 67) among the study participants. The mean age at the start of Nusinersen therapy was 36 years (15 to 69 years, ±13). The motor function of the participants at the beginning of the observational study varied greatly and was mainly determined by the type of SMA.

Figure: Age at the start of nusinersen therapy. The age at the start of treatment is shown in years. n = number of patients, p = significance value, ᴑ = mild outliers
The treatment goals of Nusinersen therapy are individualized
The following areas were named by the participants as individual treatment goals of Nusinersen therapy: Head control 4.4% (n = 13), speech or swallowing function 5.4% (n = 16), arm function 22.0% (n = 65), respiratory function 5.1% (n = 15), trunk function 11.5% (n = 76), leg function 25,7 % (n = 76), of which walking 7.1 % (n = 21), standing 1.7 % (n = 5), climbing stairs 4.7 % (n = 14), general symptoms 26.0 % (n = 77), of which generalized weakness n = 54, pain n = 14.
Majority of participants achieve an improvement in target symptoms
At the time of the first data collection, a mean symptom severity of all target symptoms of 3.7 MYMOP scale points (MYMOP – Measure Yourself Medical Outcome Profile) was described. In the course of Nusinersen therapy, the symptom severity decreased to 3.3 MYMOP scale points (10 % relative decrease in symptom severity, p < 0.001). A significant symptom improvement was achieved in head control (ability to hold the head upright and stabilize it) as well as in speech and swallowing function (decrease of 1.3 scale points, 37% relative decrease in symptom severity, p = 0.004).
In the overall cohort, 64% of all people with SMA (n = 59) showed an improvement in at least one of two target symptoms, which by definition were classified as “responders” to Nusinersen therapy. In the group of “responders”, 24 people (28% of the total cohort) showed an improvement in both individual target symptoms. Only one participant (1.1%) showed a deterioration in both target symptoms and activity and is therefore classified as a “non-responder”.
High global therapy satisfaction
Treatment satisfaction – determined by the TSQM-9 questionnaire (Treatment Satisfaction Questionnaire for Medication-9) – showed a high level of satisfaction with the efficacy of nusinersen. User-friendliness was rated lower. Overall satisfaction was again high – 95% overall were convinced that Nusinersen therapy was “good for them” (n = 132).

Figure: Overall satisfaction or dissatisfaction with the drug nusinersen. This assessment was determined using the TSQM-9: “Overall, how satisfied or dissatisfied are you with this medication?”; n = number of patients.
Satisfaction increases over the course of treatment
Satisfaction was examined on the basis of Nusinersen’s likelihood to recommend – determined by the NPS value (Net Promoter Score). At 63.6 % (n = 89), the likelihood of recommendation showed a high proportion of “promoters” (patients with a strong likelihood of recommendation), while the patient group of “detractors” (patients who do not recommend Nusinersen) was remarkably low at 12.8 % (n = 18). The NPS showed an increase in the likelihood of recommendation depending on the duration of nusinersen therapy (therapy duration of 12 months: NPS +37; 12 to 24 months: NPS +45; more than 24 months: NPS +66).
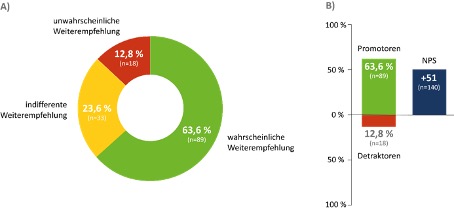
Figure: The probability of recommendation was measured by the Net Promoter Score (NPS): unlikely to recommend (0-6 points); indifferent recommendation (7-8 points); likely to recommend (9-10 points).
Positive perception of nusinersen in 5q-SMA in adulthood
In adults with 5q-SMA, nusinersen has been used across a wide age range. Treatment was initiated with a wide range of disease duration and motor-functional deficits. The treatment goals of Nusinersen therapy are individualized and relate to precisely defined symptomatic areas. The target symptoms were defined by the patients themselves and quantified in terms of symptom severity.
An improvement in the target symptoms was achieved in the majority of participants. Overall treatment satisfaction with Nusinersen was high and the treatment discontinuation rate was very low, although the user-friendliness of the treatment was rated more critically. Satisfaction with Nusinersen was exceptionally high, as determined by the probability of recommending the drug to others. Overall, the results of our application study show a positive perception of Nusinersen therapy among people with 5q-SMA who have received treatment in adulthood.
Thanks for your participation and data donation
We would like to thank all the people who took part in our observational study and provided their data for the research. The research project could only succeed thanks to their support and data donation.
Your contact for questions about the study
Susanne Spittel, M.Sc.
Medical research
s.spittel@ambulanzpartner.de
Tel. 030-81031410