Nusinersen (Spinraza®) is approved for the treatment of 5q-associated spinal muscular atrophy (SMA). For infants and children, nusinersen can halt the progression of the disease and improve muscle function. For adults, no systematic data on treatment with nusinersen are available to date. Scientific treatment documentation is therefore necessary to improve the data situation for adults with nusinersen therapy.
A very effective way of documenting treatment is a structured evaluation of treatment on the APST care portal by the patient themselves. Three questionnaires for the digital recording of treatment with nusinersen on Ambulanzpartner are provided for the self-assessment of treatment. The documentation is designed as a scientific register study called digitNusinersen1.
Participation requires the regular completion of three questionnaires (ALSFRS-ex, TSQM and MYMOP), which are presented below.
Extended ALS function scale ALSFRS-ex
The individual progression of SMA is well recorded using the extended ALS Functional Rating Scale (ALSFRS-ex). The ALSFRS-ex questionnaire determines current impairments of everyday abilities as a result of SMA. The questionnaire consists of 15 questions on physical functions (e.g. speaking, swallowing, walking). There are 5 options for each question to assess whether an impairment is present and how severe it is. The questionnaire should be completed at regular intervals of 3 months during Nusinersen therapy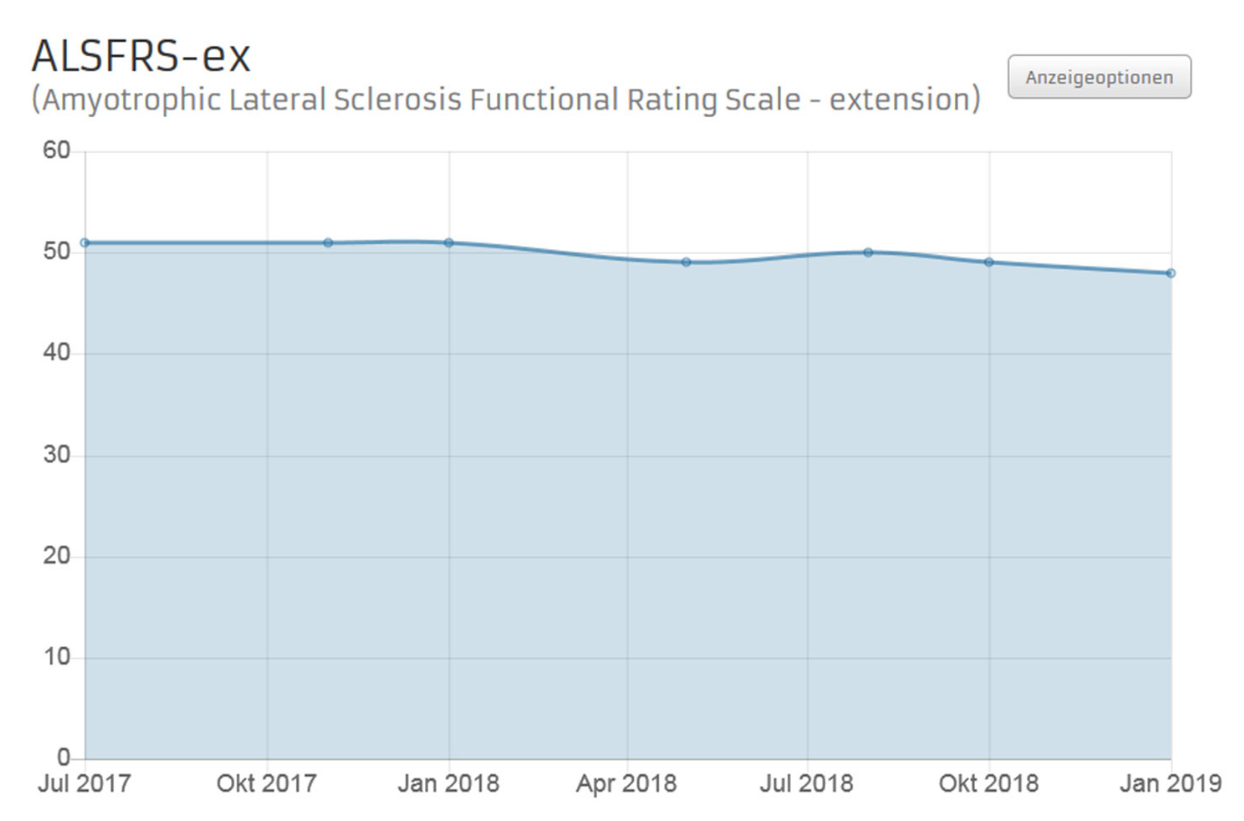
Fig 1 Example of an ALSFRS-ex progress chart in the outpatient partner patient account
Treatment satisfaction questionnaire TSQM
Satisfaction with the drug nusinersen is determined using the TSQM (Treatment Satisfaction Questionnaire for Medication) questionnaire. This is a rating scale with nine questions to record patient satisfaction with Nusinersen. The TSQM questions relate to three areas: Efficacy (questions 1 to 3), Ease of Use (questions 4 to 6) and Overall Satisfaction (questions 7 and 9). The questions are answered on a scale from very dissatisfied to very satisfied. Each of the questions is evaluated as a total value, which can take values between 0 and 100. A higher total value corresponds to a higher level of approval.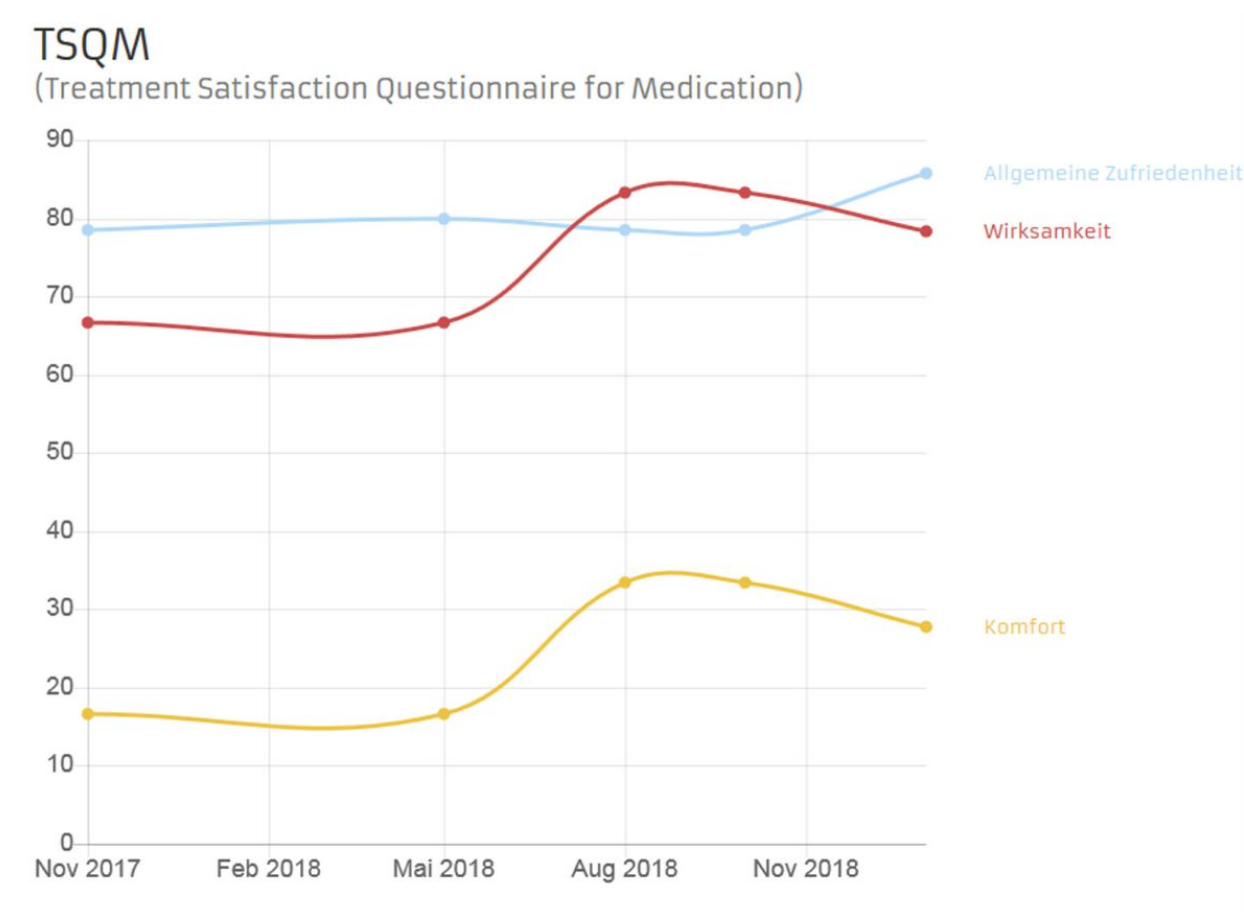
Fig. 2 Example of a progression diagram of the TSQM questionnaire in the outpatient partner patient account
The MYMOP treatment outcome questionnaire
The MYMOP (Measure Yourself Medical Outcome Profile) questionnaire is a patient-generated measurement tool. This means that the patient decides which impairments are measured. The MYMOP consists of an initial questionnaire and a follow-up questionnaire. In the first MYMOP questionnaire, the patient determines which symptom is most important to them. This symptom is entered in the questionnaire. Entering a second symptom is optional and useful but not absolutely necessary. The patient also selects an important activity of daily life that is prevented or impaired by symptom 1 or 2. The symptoms and the activity are rated on a scale of 0 to 6 according to the patient’s condition in the last week.
In the MYMOP follow-up questionnaire, the only new information is contained in a third symptom, which is optional. The MYMOP follow-up questionnaires are collected at regular intervals of 3 months.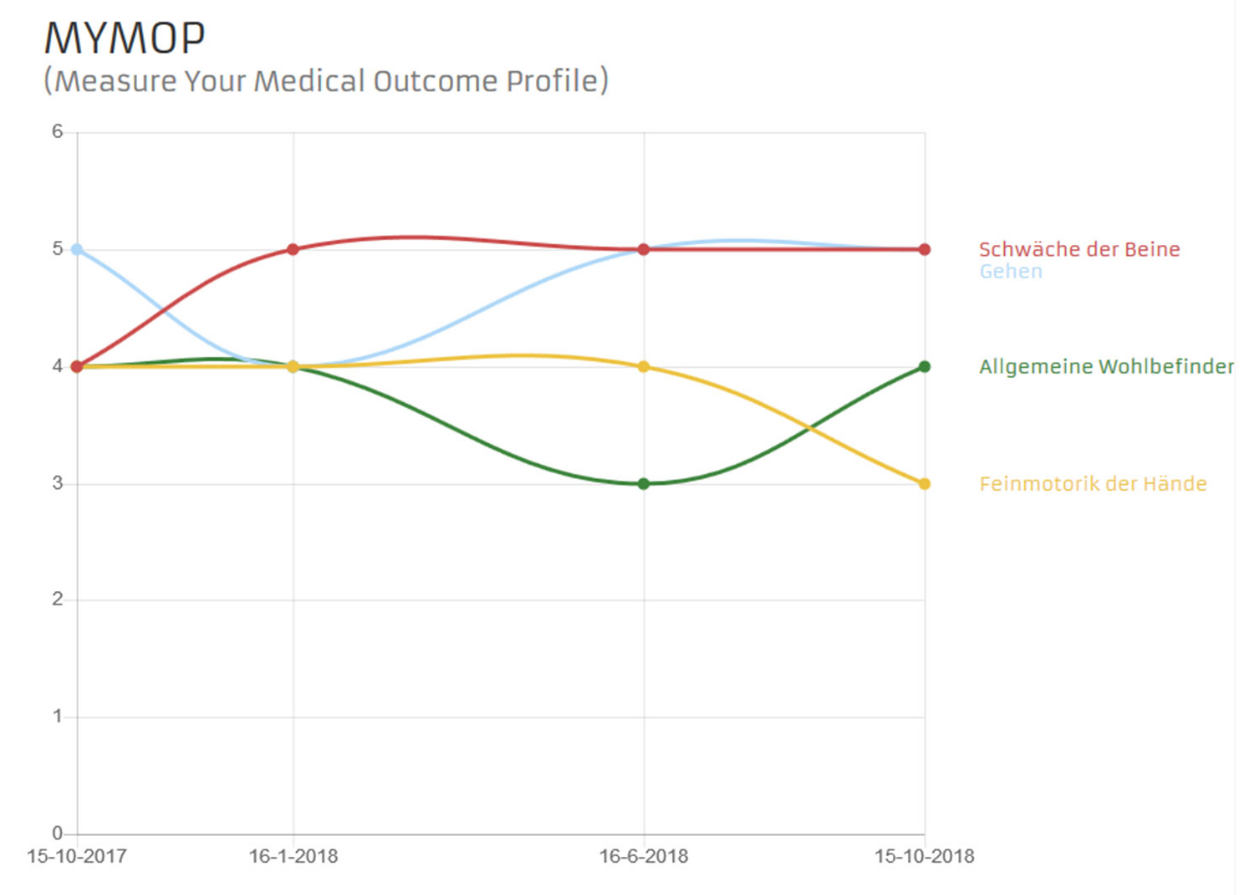
Fig. 3 Example of a progression diagram of the MYMOP questionnaire in the outpatient partner patient account
Data donation
The three questionnaires can be completed online, by telephone or in writing. The online questionnaire can be completed from home via the patient account on Ambulanzpartner. Individual appointments are made with an experienced interviewer for the telephone survey of the questionnaires. The written questionnaires are completed directly in the participating outpatient clinics.
Progress charts on ambulance partners
The results of the three questionnaires are displayed graphically as progression charts in the patient account on Ambulanzpartner. This provides patients and treating physicians with a visual representation of the course of the disease and an assessment of treatment satisfaction and outcome. These progression charts can be very useful for those involved in the complex therapy with Nusinersen.
Fig. 4 Navigation bar with the link to the ALSFRS-ex, TSQM and MYMOP progress charts in the outpatient partner patient account
Health services research
The results of the project are of great importance for research into nusinersen. The anonymized evaluation data will be made available to academic research institutions and research-based companies and will help to answer questions about care and treatment with nusinersen.
Contact us
Project digitNusinersen1
Outpatient partner care research
Westhafenstr. 1
13353 Berlin