During the course of their disease, around 90% of patients with ALS develop speech or swallowing difficulties – so-called bulbar symptoms. Around a third of all patients already show bulbar symptoms at the onset of ALS. Dextromethorphan/quinidine (DMC) has established itself as an off-label treatment option to alleviate these symptoms in ALS.
In a patient survey on DMC, we obtained experiences of people with ALS who were treated with DMC to reduce speech or swallowing difficulties. This was supplemented by a systematic analysis of patient ALSFRS self-assessment data and data from the ALS pharmacy program on DMC provision.
What are the most important study results?
Good acceptance of treatment
Patients treated with DMC took the medication for an average of 8.5 months. Around a third of those affected even used DMC for longer than nine months (Figure 1).
The treatment duration of more than eight months indicates that DMC is well accepted by many patients for the symptomatic treatment of bulbar symptoms.

Figure 1: DMC treatment duration – analysis of DMC treatment data from 768 patients with ALS. n=number of patients
High satisfaction with DMC treatment
Overall, most patients were satisfied with DMC for the treatment of their bulbar symptoms in ALS (Figure 2).
A high level of treatment satisfaction was found – measured using the “Treatment Satisfaction Questionnaire for Medication” (TSQM-9). 80% of the patients surveyed were satisfied with their treatment. Around 20 % of participants expressed some dissatisfaction or were uncertain in their assessment.

Figure 2: Satisfaction with DMC treatment – evaluation of question 9 “Overall, how satisfied or dissatisfied are you with the drug DMC?” of the questionnaire on satisfaction with drug treatment (TSQM-9). n=number of patients
Frequent recommendation of the DMC treatment
Half of the patients surveyed would recommend DMC treatment to others (50%), while some respondents were rather reluctant (27%) and others were neutral about DMC treatment (23%).
A closer look at the willingness to recommend DMC revealed a differentiated view of DMC. Most respondents with a slower (NPS +32) or medium (NPS +13) progression of ALS would recommend DMC to others. Participants with faster ALS showed a lower willingness to recommend DMC treatment (NPS +4). Nevertheless, almost half of the respondents with faster ALS (44%) would recommend the medication to other patients (Figure 3).
It cannot be excluded that patients with faster ALS progression have other disease mechanisms that are associated with a lower efficacy of DMC. Another medical explanation is that the benefit of DMC is outweighed by the progression of bulbar symptoms in faster ALS.
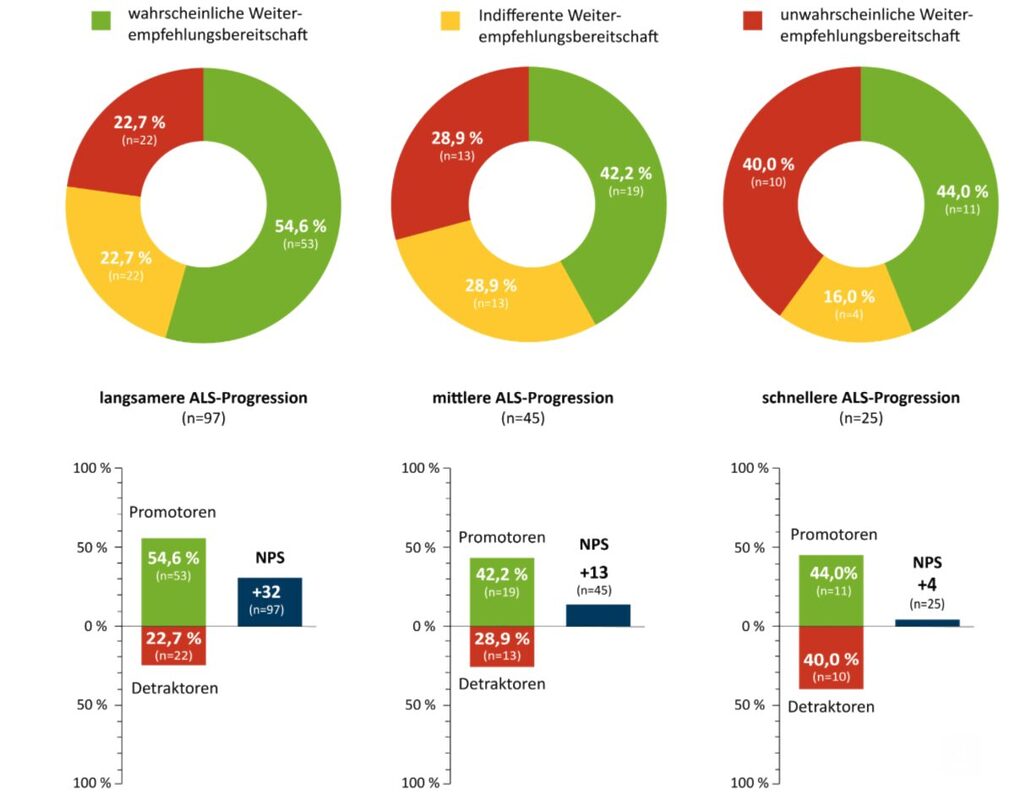
Figure 3: Willingness to recommend DMC treatment in relation to ALS progression – evaluation of willingness to recommend DMC treatment using the Net Promoter Score (NPS): unlikely to recommend (0 to 6 points “detractors”); indifferent recommendation (7 to 8 points “indifferents”); likely recommendation (9 to 10 points “promoters”). The NPS is calculated as the difference between “promoters” (proportion of promoters in the total group of patients surveyed in %) and “detractors” (proportion of detractors in the total group of patients surveyed in %). n=number of patients
How was the study conducted?
For the study, people with ALS who received the DMC medication via the ALS pharmacy program were invited to take part in a survey. The survey comprised two questionnaires:
- Net Promoter Score (NPS): Measurement of willingness to recommend DMC treatment to others
- Treatment Satisfaction Questionnaire for Medication (TSQM-9): Measurement of satisfaction with DMC treatment
Patients were able to complete the questionnaires via the ALS app.
In addition, demographic and clinical data as well as data from the patients’ ALSFRS self-assessment, which was collected using the self-explanatory ALS Functional Scale (ALSFRS-R-SE) via the ALS app, were also included.
This study is the world’s most comprehensive analysis of DMC treatment for ALS. Patients from 13 ALS centers in Germany took part in the survey. A total of 552 ALS patients receiving DMC were invited to participate in the survey; 179 patients took part in the survey (Figure 4).
The results of this study were published in a scientific journal following independent peer review. You can find the complete publication here.
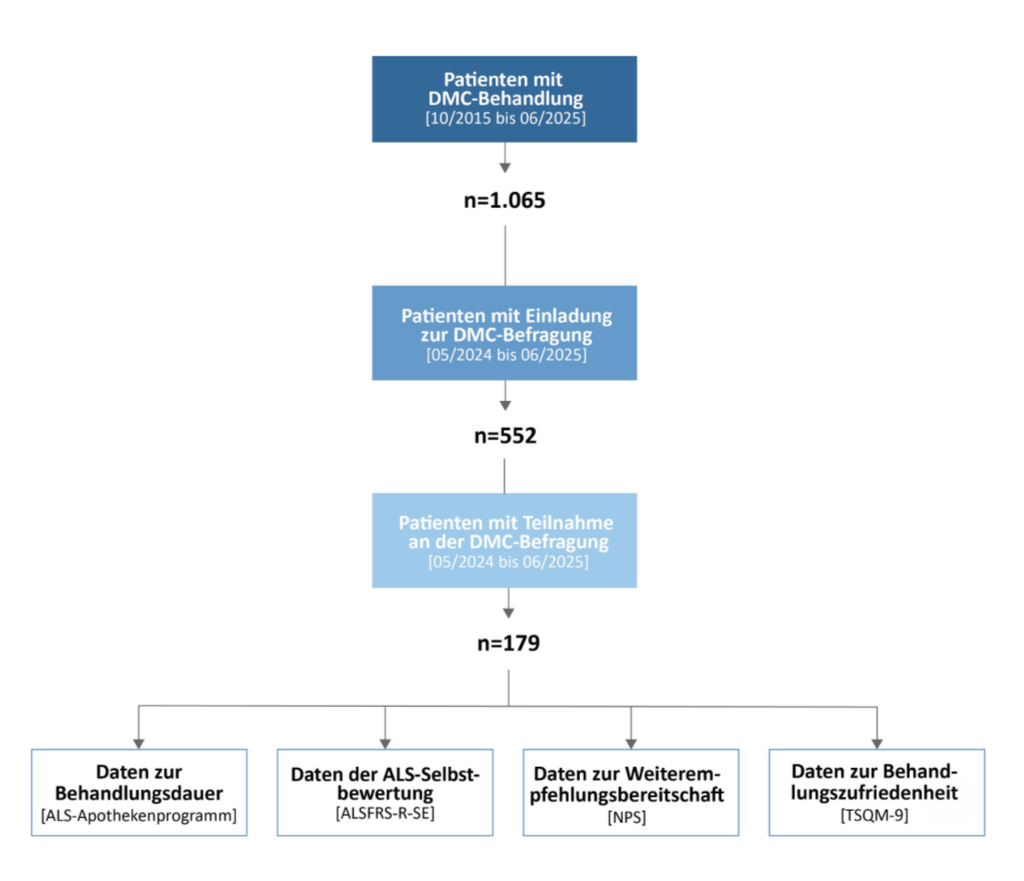
Figure 4: Study process – data collection over a period of 13 months (May 2024 – June 2025). During the study period, 552 people with ALS and DMC treatment were invited to take part in the DMC satisfaction survey. A total of 179 patients completed the online satisfaction survey via the ALS app. Data sets on treatment duration, treatment recommendation (Net Promoter Score, NPS) and treatment satisfaction (Treatment Satisfaction Questionnaire for Medication, TSQM-9) were systematically evaluated. n = number of patients with analyzable data sets.
Thanks for donating data
We would like to thank all patients who took part in this study and provided their data for research purposes. The research project could only succeed with your support and data donation.
What happens next?
We want to continue to systematically collect data on DMC treatment and bulbar symptoms in ALS.
We will make two further surveys available for this purpose:
– Questionnaire for recording bulbar symptoms – “BFS – Bulbar Function Scale”
– Questionnaire for recording ALS-typical symptoms – “ALS Symptom Scale (ALS-SYS-26)”
We will invite you to take part in our surveys by e-mail. You can take part in our surveys via the ALS app or your APST account. You can find all the questionnaires in the ALS app via the “More” tab. In your APST account, you can find all the questionnaires via the “Ratings” tab in the left-hand navigation column.
Furthermore, your regular ALSFRS self-assessment by completing the ALS Functional Scale via the ALS app is very valuable.
Thank you for your continued willingness to participate in our surveys and for your regular ALSFRS self-assessment. Your participation makes an important contribution to ALS research.
Your contact persons

How to reach us
Phone: 030 – 810 314 10
Mail: forschung@ambulanzpartner.de
Best regards
Your APST team

