APST wants to develop the biomarker neurofilament light chain (NF-L) as a criterion for the urgency of care for people with ALS. The NF-L level in the blood - in addition to ALS symptoms - is a marker of the rate of ALS progression. A high serum NF-L level is often associated with a higher rate in the progression of ALS symptoms (progression).
Consideration of a higher NF-L level should support the medical urgency of care and allow for the initiation of timely ancillary care based on the individual's disease progression. The inclusion of the biomarker NF-L in the estimation of ALS progression and decision making of an assistive care is novel and part of a research program realized by APST in collaboration with the Center for ALS and other Motor Neuron Disorders, Department of Neurology, Charité and other specialized ALS centres.
Why do we need a criterion for urgency of care?
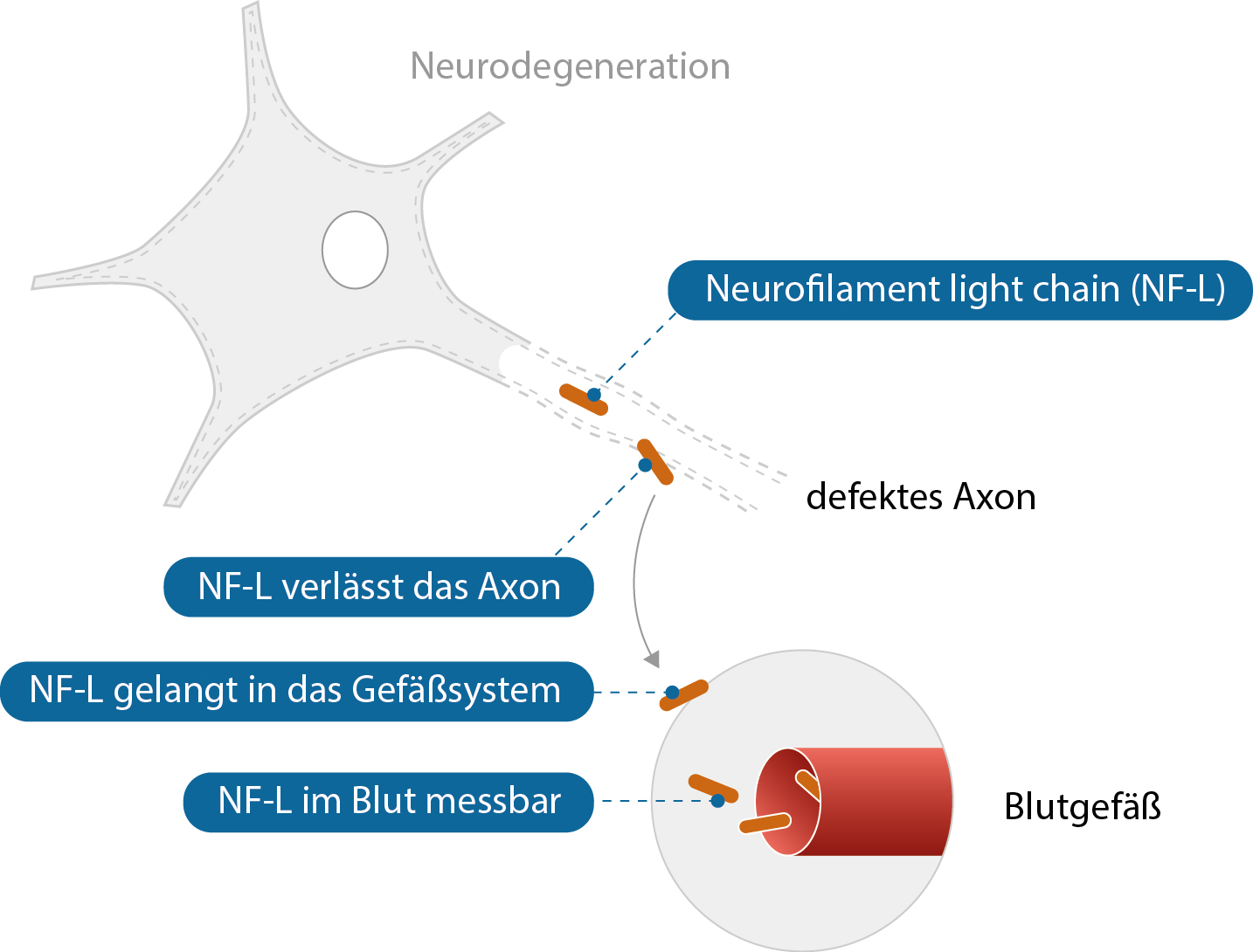
A major problem in the care of people with ALS, especially in the supply of assistive devices, is the high latency of care (the delay from the identification of a need for an assistive device to the provision of the device) (1,2). In the current care processes, especially in the approval processes at the health insurance companies, no distinction is made between fast and slow disease progression. As a result, people with high ALS progression often experience excessive delays in the provision of aids. With this in mind, we are investigating the use of the NF-L score as a criterion for urgency of care to support timely initiation of care.
What is the NF-L biomarker?
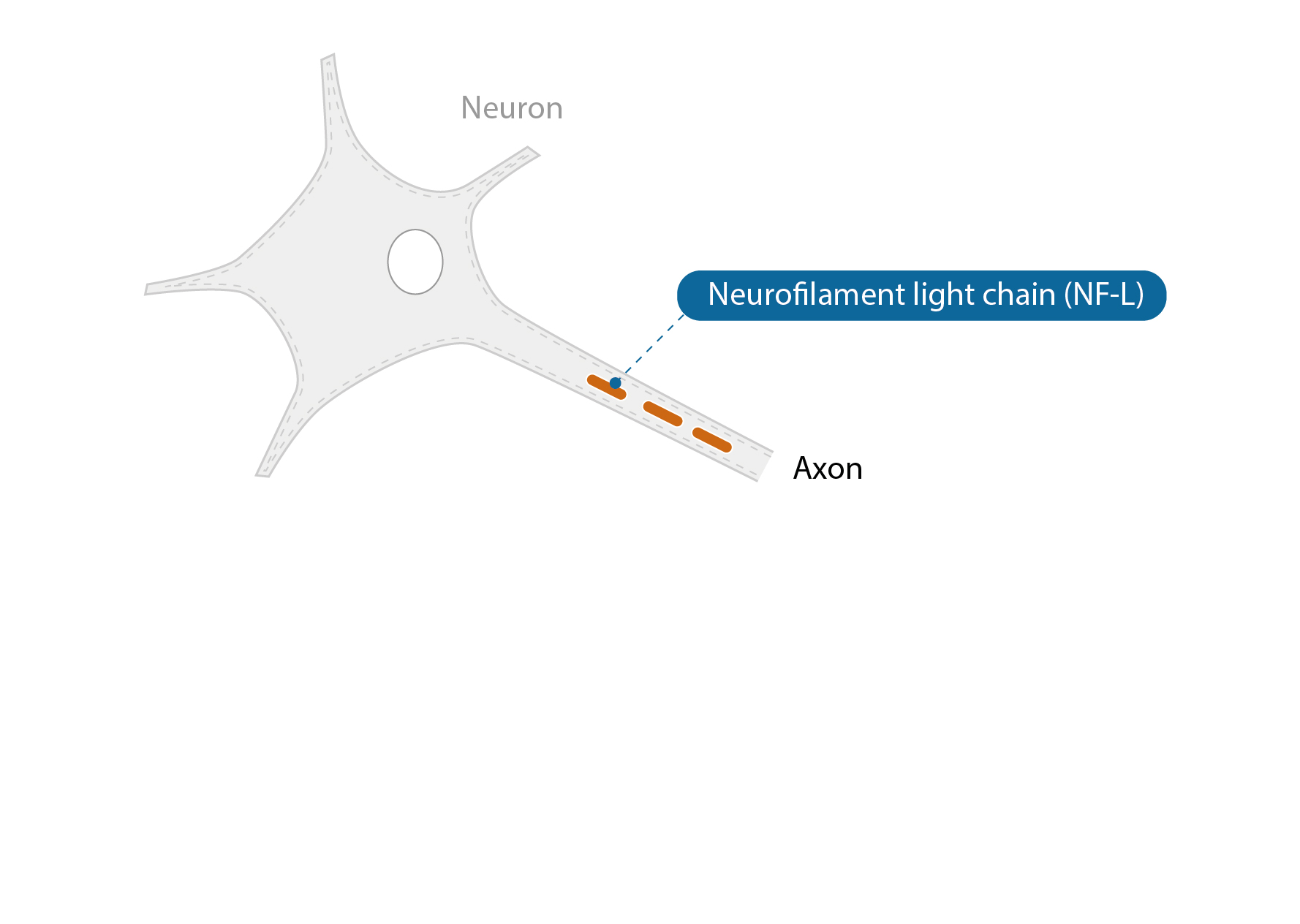
NF-L is a nerve cell-specific protein that is released into the blood when the nerve cell is damaged. NF-L is currently being investigated as a prognostic and progression marker of ALS in the "NF-L in ALS" study in Germany. Thereby, a correlation between the NF-L concentration in the blood and the prognosis of the ALS course is suspected.
How can I participate in research of NF-L as an urgency criterion for care?
The research into NFL as a criterion of care is currently being done at the ALS Center of the Charité. In the future, it is planned to include other specialized ALS centres. Participation in this project is possible if the following requirements are met:
1) Participation in the scientific study "NF-L in ALS" (Participation in the Biomarker Study NF-L in ALS.pdf).
2) Consent to participate in the APST Registry Study (Consent to the Outpatient Partner Registry Study.pdf); and
3) Consent to use data from the APST platform (Participation in Care Coordination and Use of Data for Research.pdf). Participation in NF-L includes collection of a blood sample (10 ml) from the vein twice per year.
How can the scientific data be used to improve care?
At the ALS Study Center of the Charité, Berlin, serum concentrations of NF-L are used and scientifically evaluated as an additional decision criterion for the urgency of care. In principle, a higher NF-L value indicates a higher urgency of care. The established decision-making criteria for initiating care (with medical aids and remedies, respiratory aids and nutritional therapy) remain unchanged.
In addition to the clinical examination findings, the ALS Functional Scale (ALS-FRS), the change in ALS-FRS values per month (ALS progression rate), the respiratory and weight parameters as well as various socio-medical criteria and the previous most important decision criteria. With your consent to the APST Registry Study and registration with the APST care portal, the history of your NF-L values is available for you to view in your user account on the APST Portal.
Sources:
(1) Funke A et al. Assistive device care in amyotrophic lateral sclerosis. Analysis from 3 years of case management in an internet-supported care network. The Neurologist 2015; 86: 1007-1017.
(2) Funke A et al. Provision of assistive technology devices among people with ALS in Germany: a platform-case management approach. Amyotroph Lateral Sclerosis, Frontotemporal Degener 2018;19(5-6):342-350.

.pschumann@ambulanzpartner.de
Phone 030-81031410
Mobil 0151-17639910
